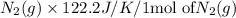Chemistry, 03.03.2020 06:05 Frenchfries13
Consider the reaction: N2(g) + 2O2(g)2NO2(g) Using standard absolute entropies at 298K, calculate the entropy change for the system when 1.90 moles of N2(g) react at standard conditions. S°system = J/K Submit Answer

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2
You know the right answer?
Consider the reaction: N2(g) + 2O2(g)2NO2(g) Using standard absolute entropies at 298K, calculate th...
Questions


Mathematics, 22.07.2021 14:00

English, 22.07.2021 14:00

Mathematics, 22.07.2021 14:00


Business, 22.07.2021 14:00


Computers and Technology, 22.07.2021 14:00


History, 22.07.2021 14:00






Business, 22.07.2021 14:00


Mathematics, 22.07.2021 14:00

Social Studies, 22.07.2021 14:00

Mathematics, 22.07.2021 14:00

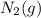 is 191.6 J/mol K,
is 191.6 J/mol K, 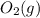 = 205 J/mol K, and
= 205 J/mol K, and 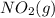 is 239.7 J/mol K at 298 K.
is 239.7 J/mol K at 298 K.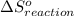 from standard absolute entropies as follows.
from standard absolute entropies as follows.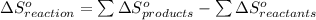
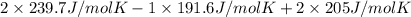
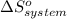 = 1.90 moles of
= 1.90 moles of