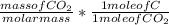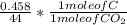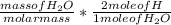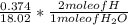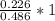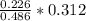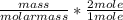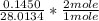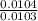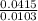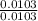Chemistry, 05.03.2020 00:53 hbhdnjdndj1867
Dimethylhydrazine is a carbon-hydrogen-nitrogen compound used in rocket fuels. When burned in an excess of oxygen, a 0.312 gg sample yields 0.458 gg CO2CO2 and 0.374 gg H2OH2O. The nitrogen content of a 0.486 gg sample is converted to 0.226 gg N2N2.What is the empirical formula of dimethylhydrazine?Express your answer as a chemical formula.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:10
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2

Chemistry, 22.06.2019 07:30
What is i fracture in the crust called when land move up, down or sideways
Answers: 2

Chemistry, 22.06.2019 13:00
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1

Chemistry, 23.06.2019 00:30
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3
You know the right answer?
Dimethylhydrazine is a carbon-hydrogen-nitrogen compound used in rocket fuels. When burned in an exc...
Questions

Mathematics, 09.04.2020 19:28

Mathematics, 09.04.2020 19:28






Mathematics, 09.04.2020 19:28



Social Studies, 09.04.2020 19:28

History, 09.04.2020 19:28



Social Studies, 09.04.2020 19:28



Mathematics, 09.04.2020 19:28


Mathematics, 09.04.2020 19:28