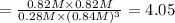Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
When an object falls through the air and encounters air resistance its overall speed will be than if it had not encountered air resistance? (one word answer)
Answers: 2

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

Chemistry, 22.06.2019 22:30
What is the value of the standard enthalpy of formation of an element in its most stable form?
Answers: 3
You know the right answer?
Carbon tetrachloride can be produced by this reaction: Suppose 1.1 mol and 3.3 mol are placed in a 1...
Questions

Social Studies, 31.07.2021 16:20




English, 31.07.2021 16:20



Social Studies, 31.07.2021 16:20

Business, 31.07.2021 16:20

Health, 31.07.2021 16:20

English, 31.07.2021 16:20


Physics, 31.07.2021 16:20

Social Studies, 31.07.2021 16:20


Mathematics, 31.07.2021 16:20




Mathematics, 31.07.2021 16:20

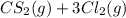 ⇌
⇌ 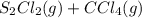
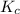 .
.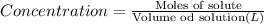
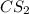 :
:![[CS_2]=\frac{1.1 mol}{1 L}=1.1 M](/tpl/images/0533/8772/cef0e.png)
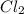 :
:![[Cl_2]=\frac{3.3mol}{1 L}=3.3M](/tpl/images/0533/8772/72054.png)
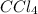 :
:![[CCl_4]=\frac{0.82 mol}{1 L}=0.82 M](/tpl/images/0533/8772/68898.png)
![K_c=\frac{[S_2Cl_2][CCl_4]}{[CS_2][Cl_2]^3}](/tpl/images/0533/8772/40ad0.png)