
Chemistry, 05.03.2020 07:01 esdancer3494
An industrial chemist studying bleaching and sterilizing prepares several hypochlorite buffers. Find the pH of 0.200 M HClO and 0.200 M NaClO. (Ka for HClO = 2.9 × 10−8)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3

Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1
You know the right answer?
An industrial chemist studying bleaching and sterilizing prepares several hypochlorite buffers. Find...
Questions


Mathematics, 01.09.2020 23:01


History, 01.09.2020 23:01

Medicine, 01.09.2020 23:01

English, 01.09.2020 23:01

Mathematics, 01.09.2020 23:01

English, 01.09.2020 23:01

Social Studies, 01.09.2020 23:01

Biology, 01.09.2020 23:01



Biology, 01.09.2020 23:01





English, 01.09.2020 23:01


Biology, 01.09.2020 23:01

![=pk_{a} +log(\frac{[conjugate base]}{[weakacid]})](/tpl/images/0534/2498/9fd53.png)
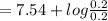 = 7.54
= 7.54

