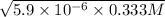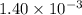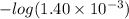Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 12:30
4nh3+5o2--> 4no+6h20what is the total number of moles of h2o produced when 12 mole of nh3 is completely consumed?
Answers: 3

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 21:30
Describe at least two advantages and two disadvantages of using hydropower as a source of energy.
Answers: 2
You know the right answer?
Consider the titration of 50.0 mL of 1.00 M C5H5N by 0.500 M HCl. For each volume of HCl added, deci...
Questions



Chemistry, 20.09.2020 05:01



Biology, 20.09.2020 05:01


English, 20.09.2020 05:01


Geography, 20.09.2020 05:01


English, 20.09.2020 05:01

Social Studies, 20.09.2020 05:01

Mathematics, 20.09.2020 05:01

Mathematics, 20.09.2020 05:01



Geography, 20.09.2020 05:01


Mathematics, 20.09.2020 05:01

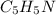 by 0.500 M HCl. For each volume of HCl added, decide which of the components is a major species after the HCl has reacted completely. Kb for
by 0.500 M HCl. For each volume of HCl added, decide which of the components is a major species after the HCl has reacted completely. Kb for 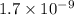 . Calculate the pH at the equivalence point for this titration.
. Calculate the pH at the equivalence point for this titration. 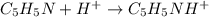
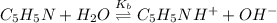
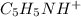 at the equivalence point. It is acidic with a pH less than 7.
at the equivalence point. It is acidic with a pH less than 7.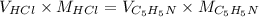
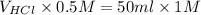
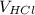 = 100 ml
= 100 ml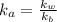
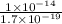
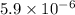
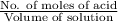
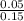
![[H^{+}] = \sqrt{k_{a} \times C}](/tpl/images/0534/4757/8d15a.png)