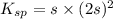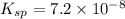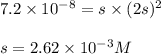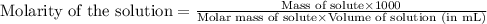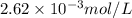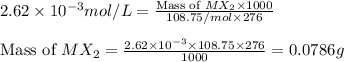Chemistry, 05.03.2020 15:09 ImmortalEnigmaYT
The solubility product constant for MX2 is 7.2 x 10-8. How many grams of MX2 (108.75 g/mol) will dissolve in 276 ml of water at 25°C. M is the metal and X is the anion. Enter as a number to 4 decimal places.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Choose all the answers that apply. ionic compounds dissolve easily in water do not dissolve in water have low melting points have high melting points conduct electricity when melted
Answers: 1

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3

Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2

Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3
You know the right answer?
The solubility product constant for MX2 is 7.2 x 10-8. How many grams of MX2 (108.75 g/mol) will dis...
Questions


Mathematics, 25.01.2021 18:50

Mathematics, 25.01.2021 18:50

Mathematics, 25.01.2021 18:50

Mathematics, 25.01.2021 18:50

Mathematics, 25.01.2021 18:50


Biology, 25.01.2021 18:50

Arts, 25.01.2021 18:50


English, 25.01.2021 18:50

History, 25.01.2021 18:50

Mathematics, 25.01.2021 18:50

Physics, 25.01.2021 18:50


Mathematics, 25.01.2021 18:50

Mathematics, 25.01.2021 18:50


Arts, 25.01.2021 18:50

Chemistry, 25.01.2021 18:50

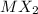 that will dissolve is 0.0786 grams
that will dissolve is 0.0786 grams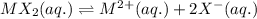
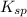 for above equation follows:
for above equation follows: