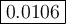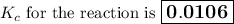Calculate the value of the equilibrium constant, K c , for the reaction Q ( g ) + X ( g ) − ⇀ ↽ − 2 M ( g ) + N ( g ) given that M ( g ) − ⇀ ↽ − Z ( g ) K c 1 = 3.15 6 R ( g ) − ⇀ ↽ − 2 N ( g ) + 4 Z ( g ) K c 2 = 0.509 3 X ( g ) + 3 Q ( g ) − ⇀ ↽ − 9 R ( g ) K c 3 = 12.5

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:50
H2so4(aq) + mg(s)—> mgso4(aq) +h2(g) which substance is the acid in the reaction?
Answers: 3


Chemistry, 22.06.2019 07:10
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2
You know the right answer?
Calculate the value of the equilibrium constant, K c , for the reaction Q ( g ) + X ( g ) − ⇀ ↽ − 2...
Questions





Mathematics, 09.11.2020 03:00


Medicine, 09.11.2020 03:00


Chemistry, 09.11.2020 03:00

Biology, 09.11.2020 03:00

English, 09.11.2020 03:00

History, 09.11.2020 03:00

History, 09.11.2020 03:00


Chemistry, 09.11.2020 03:00





History, 09.11.2020 03:00