
Chemistry, 06.03.2020 16:58 ramireztony741
Using the equations 2 Sr(s) + O₂ (g) → 2 SrO (s) ∆H° = -1184 kJ/mol SrO (s) + CO₂ (g) → SrCO₃ (s) ∆H° = -234 kJ/mol CO₂ (g) → C(s) + O₂ (g) ∆H° = 394 kJ/mol Determine the enthalpy for the reaction 2 SrCO₃ (s) → 2 Sr (s) + 2 C(s) + 3 O₂ (g).

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3


Chemistry, 22.06.2019 09:00
Ineed to find the answer of this question because i dont understand it
Answers: 1

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1
You know the right answer?
Using the equations 2 Sr(s) + O₂ (g) → 2 SrO (s) ∆H° = -1184 kJ/mol SrO (s) + CO₂ (g) → SrCO₃ (s) ∆H...
Questions


Biology, 21.10.2019 23:30

Chemistry, 21.10.2019 23:30






Arts, 21.10.2019 23:30

History, 21.10.2019 23:30

Mathematics, 21.10.2019 23:30


History, 21.10.2019 23:30

Social Studies, 21.10.2019 23:30






Mathematics, 21.10.2019 23:30

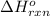 for the reaction is 72 kJ.
for the reaction is 72 kJ.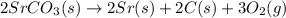
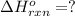
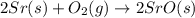
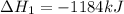
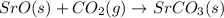
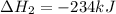 ( × 2)
( × 2)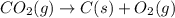
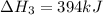 ( × 2)
( × 2)![\Delta H^o_{rxn}=[1\times (\Delta H_1)]+[2\times (-\Delta H_2)]+[2\times (\Delta H_3)]](/tpl/images/0535/5885/feb85.png)
![\Delta H^o_{rxn}=[(1\times (-1184))+(2\times -(-234))+(2\times (394))]=72kJ](/tpl/images/0535/5885/d98fd.png)


