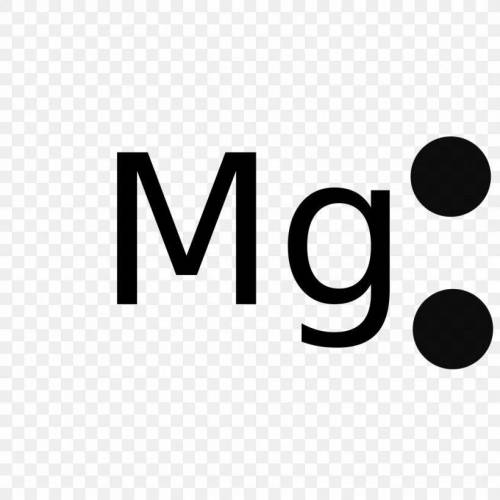Chemistry, 07.03.2020 01:25 willoughbysierra
Identify the atoms that correspond to each of the following electron configurations. Then, write the Lewis symbol for the common ion formed from each atom: (a) 1s22s22p5 (b) 1s22s22p63s2 (c) 1s22s22p63s23p64s23d10 (d) 1s22s22p63s23p64s23d104p4 (e) 1s22s22p63s23p64s23d104p1

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3


Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

You know the right answer?
Identify the atoms that correspond to each of the following electron configurations. Then, write the...
Questions

Chemistry, 16.10.2020 16:01

Computers and Technology, 16.10.2020 16:01


English, 16.10.2020 16:01

Biology, 16.10.2020 16:01



Mathematics, 16.10.2020 16:01


Mathematics, 16.10.2020 16:01








Mathematics, 16.10.2020 16:01

Mathematics, 16.10.2020 16:01