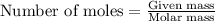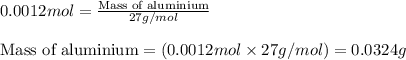Chemistry, 07.03.2020 04:38 iwantcandy2002
Aluminum reacts with excess hydrochloric acid to form aqueous aluminum chloride and 46.5 mL of hydrogen gas over water at 27 degrees Celsius and 751 mmHg. How many grams of aluminum reacted? The partial pressure of water at 27 C is 26.8 mmHg.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2

Chemistry, 22.06.2019 11:00
Surface currents are caused by blank space . question 14 options: surface currents are caused by? differences in water temperature high salinity differences in density wind forces
Answers: 1

Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 23.06.2019 09:30
What is the force of an object when it landed(sitting in the ground)
Answers: 2
You know the right answer?
Aluminum reacts with excess hydrochloric acid to form aqueous aluminum chloride and 46.5 mL of hydro...
Questions

Engineering, 18.10.2020 01:01






Mathematics, 18.10.2020 01:01




Mathematics, 18.10.2020 01:01

English, 18.10.2020 01:01


Geography, 18.10.2020 01:01

Geography, 18.10.2020 01:01


Chemistry, 18.10.2020 01:01

Physics, 18.10.2020 01:01

Mathematics, 18.10.2020 01:01

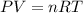
![27^oC=[27+273]K=300K](/tpl/images/0537/7176/16c81.png)
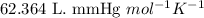
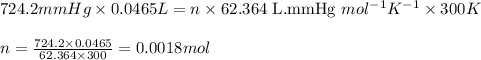
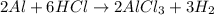
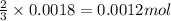 of aluminium is reacted
of aluminium is reacted