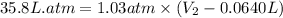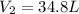Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3


Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3

Chemistry, 23.06.2019 07:00
Explain what happened when the storm surges from hurricanes reached the gulf coast
Answers: 1
You know the right answer?
A sample of gas occupies a volume of 64.0 mL . As it expands, it does 137.1 J of work on its surroun...
Questions




English, 16.12.2021 18:20





Mathematics, 16.12.2021 18:20




World Languages, 16.12.2021 18:20


Physics, 16.12.2021 18:20


Social Studies, 16.12.2021 18:20

Social Studies, 16.12.2021 18:20


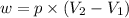
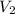 = final volume = ?
= final volume = ?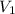 = initial volume = 64.0 mL = 0.0640 L (1 L = 1000 mL)
= initial volume = 64.0 mL = 0.0640 L (1 L = 1000 mL)