
Chemistry, 09.03.2020 02:21 imstupid77
If solution A has a pH that is four pH units greater than that of solution B, how much greater is the H3O+ concentration in solution B than in solution A?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 17:20
Which of these features are formed when hot groundwater is forced out through cracks in the earth's surface?
Answers: 2

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

You know the right answer?
If solution A has a pH that is four pH units greater than that of solution B, how much greater is th...
Questions


Mathematics, 01.08.2019 05:30




English, 01.08.2019 05:30







Biology, 01.08.2019 05:30




Biology, 01.08.2019 05:30

Mathematics, 01.08.2019 05:30


![pH = -log([H_{3}O^{+}])](/tpl/images/0538/6275/6ab72.png)
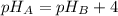
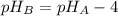
![[H_{3}O^{+}]_{B} = 10^{-pH_{B}}](/tpl/images/0538/6275/1b6e4.png)
![[H_{3}O^{+}]_{B} = 10^{-(pH_{A} - 4)}](/tpl/images/0538/6275/4b06e.png)
![[H_{3}O^{+}]_{B} = 10^{-(-log[H_{3}O^{+}]_{A} - 4)}](/tpl/images/0538/6275/1f70b.png)
![[H_{3}O^{+}]_{B} = 10^{(log[H_{3}O^{+}]_{A} + 4)}](/tpl/images/0538/6275/cd0c6.png)
![[H_{3}O^{+}]_{B} = 10^{4}[H_{3}O^{+}]_{A}](/tpl/images/0538/6275/ec01c.png)


