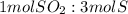Chemistry, 09.03.2020 03:20 coolkitty35
Using the above equation, how many moles of sulfur can be produced if 0.5640 moles of sulfur dioxide are used with excess hydrogen sulfide?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:00
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1


Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 15:30
The identities of substances are the same before and after which type of change
Answers: 1
You know the right answer?
Using the above equation, how many moles of sulfur can be produced if 0.5640 moles of sulfur dioxide...
Questions


Mathematics, 05.08.2021 23:40






Social Studies, 05.08.2021 23:40





Mathematics, 05.08.2021 23:40

Mathematics, 05.08.2021 23:50


Biology, 05.08.2021 23:50

History, 05.08.2021 23:50


Mathematics, 05.08.2021 23:50