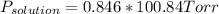Chemistry, 09.03.2020 18:50 lattimore12
Biphenyl, C12H10, is a nonvolatile, nonionizing solute that is soluble in benzene, C6H6. At 25 ∘C, the vapor pressure of pure benzene is 100.84 Torr. What is the vapor pressure of a solution made from dissolving 11.5 g of biphenyl in 31.9 g of benzene?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:30
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1


Chemistry, 22.06.2019 19:00
Imagine that a new planet is discovered with two moons of equal mass: moon a and moon b. the mass of the new planet is greater than the combined mass of its moons. moon a is farther away from the new planet than moon b. what is the planet's gravitational pull on moon a compared to the planet's gravitational pull on moon b? the planet's gravity repels moon a with a greater force than it repels moon b, which is why moon a is farther away. the gravitational pull on moon b is greater than on moon a because moon b is closer to the new planet than moon a. the gravitational pull on moon b is greater than on moon a because moon b is farther away from the new planet than moon a. the gravitational pull on moon a is the same as the gravitational pull on moon b because distance does not affect the planet's gravity.
Answers: 1

Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1
You know the right answer?
Biphenyl, C12H10, is a nonvolatile, nonionizing solute that is soluble in benzene, C6H6. At 25 ∘C, t...
Questions


Mathematics, 03.02.2020 07:52


History, 03.02.2020 07:52

History, 03.02.2020 07:52

History, 03.02.2020 07:52


Mathematics, 03.02.2020 07:53

Mathematics, 03.02.2020 07:53

History, 03.02.2020 07:53


English, 03.02.2020 07:53

Chemistry, 03.02.2020 07:53


History, 03.02.2020 07:53

History, 03.02.2020 07:53


Mathematics, 03.02.2020 07:53


Mathematics, 03.02.2020 07:53


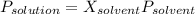 (1)
(1)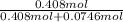 = 0.846
= 0.846