Problem Page Question An analytical chemist weighs out of an unknown monoprotic acid into a volumetric flask and dilutes to the mark with distilled water. He then titrated this solution with solution. When the titration reaches the equivalence point, the chemist finds he has added of solution. Calculate the molar mass of the unknown acid. Round your answer to significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1


Chemistry, 22.06.2019 13:30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2

Chemistry, 22.06.2019 18:00
The human activities in two locations are described below: location a: rampant use of plastic containers location b: excessive use of pesticides and fertilizers which statement is most likely true? location a will have poor air quality because plastic is biodegradable. location a will experience water scarcity because plastic absorbs moisture. the population of honeybees will increase in location b because production of crops will increase. the population of fish in location b will decrease because the water is contaminated.
Answers: 1
You know the right answer?
Problem Page Question An analytical chemist weighs out of an unknown monoprotic acid into a volumetr...
Questions

English, 20.11.2020 19:30


Arts, 20.11.2020 19:30


Mathematics, 20.11.2020 19:30

Mathematics, 20.11.2020 19:30

Mathematics, 20.11.2020 19:30

Biology, 20.11.2020 19:30

History, 20.11.2020 19:30



Mathematics, 20.11.2020 19:30

History, 20.11.2020 19:30

English, 20.11.2020 19:30


Mathematics, 20.11.2020 19:30



Mathematics, 20.11.2020 19:30

Mathematics, 20.11.2020 19:30

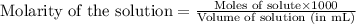
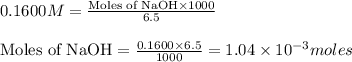

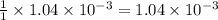 moles of HA
moles of HA