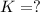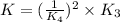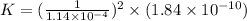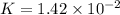Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Which of the following best supports the concept that genetic information is passed on to offspring from both of their parents, not just one?
Answers: 2


Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2
You know the right answer?
Given the two reactions PbCl2(aq)⇌Pb2+(aq)+2Cl−(aq), K3 = 1.84×10−10, and AgCl(aq)⇌Ag+(aq)+Cl−(aq),...
Questions

History, 04.12.2021 23:10

History, 04.12.2021 23:10

Biology, 04.12.2021 23:10


Mathematics, 04.12.2021 23:10


Mathematics, 04.12.2021 23:10

Mathematics, 04.12.2021 23:10

Mathematics, 04.12.2021 23:10

Chemistry, 04.12.2021 23:10


Mathematics, 04.12.2021 23:10

History, 04.12.2021 23:10

Advanced Placement (AP), 04.12.2021 23:10

Business, 04.12.2021 23:10



English, 04.12.2021 23:10


Mathematics, 04.12.2021 23:10

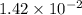
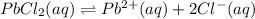 ;
; 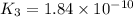
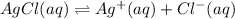 ;
; 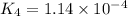
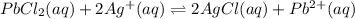 ;
;