Chemistry, 10.03.2020 06:58 nikitakhrabkov123
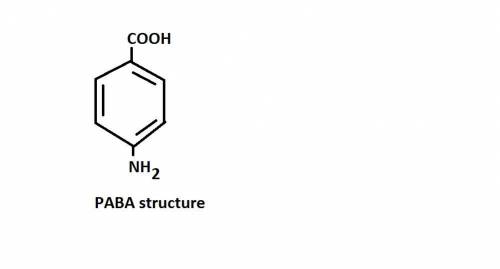
1. PABA is .
2. The molecule contains both a C O O H group and an − N H 2 group. It can act like an H +blank.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2

Chemistry, 23.06.2019 02:00
When an experimenter draws a conclusion that he assumes will apply to all situations set up similarly to his test situation, even though he cannot possibly have examined all possible test scenarios, the experimenter is using deductive reasoning inductive reasoning abductive reasoning subjective reasoning
Answers: 1

Chemistry, 23.06.2019 13:30
Explain the impact that changing the temperature has on a system in a state of dynamic equilibrium. what will happen when the temperature of an exothermic reaction mixture at equilibrium is increased?
Answers: 3
You know the right answer?
1. PABA is .
2. The molecule contains both a C O O H group and an − N H 2 group. It can act...
2. The molecule contains both a C O O H group and an − N H 2 group. It can act...
Questions

Mathematics, 19.09.2021 05:30


English, 19.09.2021 05:30

History, 19.09.2021 05:30




Social Studies, 19.09.2021 05:30

Mathematics, 19.09.2021 05:30


Biology, 19.09.2021 05:30


Social Studies, 19.09.2021 05:30

Mathematics, 19.09.2021 05:30

Mathematics, 19.09.2021 05:30


Mathematics, 19.09.2021 05:30


Mathematics, 19.09.2021 05:30

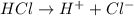
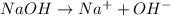
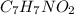 . This compound can take up a proton and also it is able to lose a proton.
. This compound can take up a proton and also it is able to lose a proton. 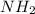 contains a lone pair of electrons and it is difficult for it to lose a hydrogen ion but it can readily gain a hydrogen ion. Therefore, this group acts as a base.
contains a lone pair of electrons and it is difficult for it to lose a hydrogen ion but it can readily gain a hydrogen ion. Therefore, this group acts as a base.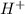 donor or acceptor.
donor or acceptor.