
50.0 mL solution of 0.160 M potassium alaninate ( H 2 NC 2 H 5 CO 2 K ) is titrated with 0.160 M HCl . The p K a values for the amino acid alanine are 2.344 ( p K a1 ) and 9.868 ( p K a2 ) , which correspond to the carboxylic acid and amino groups, respectively. a) how do you calculate the PH of the first equivalent and b) the second equivalent? please help

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
In the 1800s, one of the statements in john dalton's atomic theory was that atoms are indivisible. later experimental evidence led to the discovery of subatomic particles such as neutrons, electrons, and protons. what happened to the indivisible atom part of dalton's atomic theory, and why?
Answers: 3

Chemistry, 22.06.2019 07:30
Plz mark brainliest 30 points 1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s. 2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 14:00
650.j is the same amount of energy as? 2720cal1550cal650.cal2.72cal
Answers: 2
You know the right answer?
50.0 mL solution of 0.160 M potassium alaninate ( H 2 NC 2 H 5 CO 2 K ) is titrated with 0.160 M HCl...
Questions


Mathematics, 16.12.2021 01:50

Social Studies, 16.12.2021 01:50



Computers and Technology, 16.12.2021 01:50

Mathematics, 16.12.2021 01:50


Biology, 16.12.2021 01:50

Mathematics, 16.12.2021 01:50



History, 16.12.2021 01:50

Social Studies, 16.12.2021 01:50


Advanced Placement (AP), 16.12.2021 01:50



Social Studies, 16.12.2021 01:50

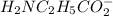

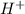 ------>
------> 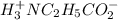
![[H_3}^+NC_2H_5CO^-_2]](/tpl/images/0540/7336/f75f8.png) =
= 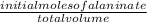
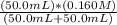
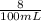
![[H^+]](/tpl/images/0540/7336/07acb.png) =
= ![\sqrt{\frac{K_{a1}K_{a2}{[H_3}^+NC_2H_5CO^-_2]+K_{a1}K_w}{ K_{a1}{[H_3}^+NC_2H_5CO^-_2] } }](/tpl/images/0540/7336/a0e17.png)
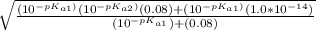

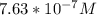
![-log[7.63*10^{-7}]](/tpl/images/0540/7336/a344f.png)
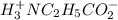
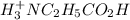
![[H^+_3NC_2H_5CO_2H]](/tpl/images/0540/7336/4ff0a.png) =
= 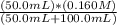
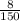
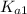 =
= ![\frac{[H^+] [H^+_3NC_2H_5CO^-_2]}{[H^+_3NC_2H_5CO_2H]}](/tpl/images/0540/7336/e2bce.png)
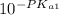 =
= 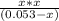
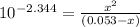
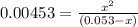
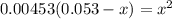
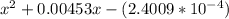
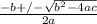
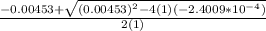 OR
OR 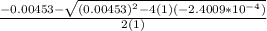
![[H^+]=[H_3^+NC_2H_5CO^-_2]= 0.0134 M](/tpl/images/0540/7336/73fad.png)
![-log[H^+]](/tpl/images/0540/7336/cbdd4.png)
![-log[0.0134]](/tpl/images/0540/7336/6b2f8.png)


