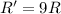Chemistry, 10.03.2020 19:06 ryliekarenbowers
By what factor does the rate change in each of the following cases (assuming constant temperature)?
Factor (enter as decimal if <1)
(a) A reaction is first order in reactant A, and [A] is doubled.
(b) A reaction is second order in reactant B, and [B] is halved.
(c) A reaction is second order in reactant C, and [C] is tripled.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
How many moles are in 7.2 x 10^23 carbon molecules? (*round to the nearest hundredth and include the unit "mol c" after your number) question 6 options:
Answers: 2

Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1

Chemistry, 22.06.2019 16:00
No copying 15 pts how does a free-body diagram tell you about the net force on an object?
Answers: 2

Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1
You know the right answer?
By what factor does the rate change in each of the following cases (assuming constant temperature)?...
Questions





Computers and Technology, 05.10.2021 07:40


Biology, 05.10.2021 07:40

Physics, 05.10.2021 07:40

Physics, 05.10.2021 07:40


English, 05.10.2021 07:40


Arts, 05.10.2021 07:40

English, 05.10.2021 07:40



Arts, 05.10.2021 07:40


Chemistry, 05.10.2021 07:40

Chemistry, 05.10.2021 07:40

![R=k[A]^1](/tpl/images/0541/2780/9975f.png)
![R'=k[2A]^1](/tpl/images/0541/2780/53afe.png)
![R'=2k[A]](/tpl/images/0541/2780/37f63.png)
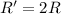
![R=k[B]^2](/tpl/images/0541/2780/92409.png)
![R'=k[\frac{B}{2}]^2](/tpl/images/0541/2780/d1975.png)
![R'=\frac{1}{4}k[B]^2](/tpl/images/0541/2780/1571d.png)
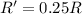
![R=k[C]^2](/tpl/images/0541/2780/01b2f.png)
![R'=k[3C]^2](/tpl/images/0541/2780/92d86.png)
![R'=9k[C]^2](/tpl/images/0541/2780/6f960.png)