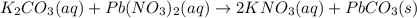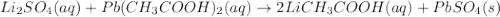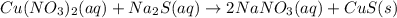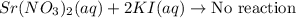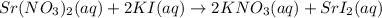Write a molecular equation for the precipitation reaction that occurs (if any) when the following solutions are mixed. If no reaction occurs, write NOREACTION.
Part A
potassium carbonate and lead(II) nitrate
Express your answer as a chemical equation. Enter NOREACTION if no reaction occurs. Identify all of the phases in your answer.
Part B
lithium sulfate and lead(II) acetate
Express your answer as a chemical equation. Enter NOREACTION if no reaction occurs. Identify all of the phases in your answer.
Part C
copper(II) nitrate and sodium sulfide
Express your answer as a chemical equation. Enter NOREACTION if no reaction occurs. Identify all of the phases in your answer.
Part D
strontium nitrate and potassium iodide
Express your answer as a chemical equation. Enter NOREACTION if no reaction occurs. Identify all of the phases in your answer.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Chemistry, 23.06.2019 01:30
Ariver current has a velocity of 5km/h relative to the shore, and a boat moves in the same direction as the current at 5 km/h relative to the river. how can the velocity of the boat relative to the shore be calculated?
Answers: 1

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1
You know the right answer?
Write a molecular equation for the precipitation reaction that occurs (if any) when the following so...
Questions


Social Studies, 17.06.2021 14:00


Mathematics, 17.06.2021 14:00

Biology, 17.06.2021 14:00


Business, 17.06.2021 14:00

Computers and Technology, 17.06.2021 14:00


Spanish, 17.06.2021 14:00


English, 17.06.2021 14:00

Biology, 17.06.2021 14:00

History, 17.06.2021 14:00


History, 17.06.2021 14:00


English, 17.06.2021 14:00

History, 17.06.2021 14:00

Chemistry, 17.06.2021 14:00