
Chemistry, 10.03.2020 19:00 baileyanne9389
What volume (in L) of oxygen will be required to produce 77.4 L of water vapor in the reaction below?
2c2H6(g)+7O2(g)--->4CO2(g)+6H2O( g)
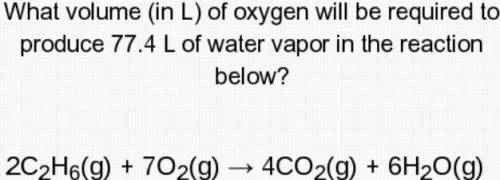

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Which statement best explains the relationship between an area is geography and the temperature of its surface water
Answers: 1

Chemistry, 22.06.2019 16:00
Which process transfers heat from inside earth to its surface? convection currents in mantle pulling away of tectonic plates drawing in of tectonic plates convection currents in crust
Answers: 1

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2

Chemistry, 23.06.2019 01:30
What happens to the concentration of hydronium ions as the ph of a solution increases? a. hydronium ion concentration stays the same b. hydronium ion concentration decreases c. hydronium ion concentration increases
Answers: 1
You know the right answer?
What volume (in L) of oxygen will be required to produce 77.4 L of water vapor in the reaction below...
Questions


Spanish, 23.02.2021 01:10



Mathematics, 23.02.2021 01:10

Mathematics, 23.02.2021 01:10


Computers and Technology, 23.02.2021 01:10


Arts, 23.02.2021 01:10



Business, 23.02.2021 01:10


Chemistry, 23.02.2021 01:10


Mathematics, 23.02.2021 01:10





