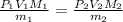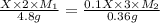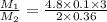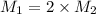Suppose you are given two flasks at the same temperature, one of volume 2 L and the other of volume 3 L. The 2-L flask contains 4.8 g of gas, and the gas pressure is X atm. The 3-L flask contains 0.36 g of gas, and the gas pressure is 0.1X. Do the two gases have the same molar mass? If not, which contains the gas of higher molar mass?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 22.06.2019 23:00
In which region is the substance in both the solid phase and the liquid phase? 1 2. 3 4 mark this and return save and exit next
Answers: 2

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1
You know the right answer?
Suppose you are given two flasks at the same temperature, one of volume 2 L and the other of volume...
Questions

Mathematics, 15.02.2021 21:00





Computers and Technology, 15.02.2021 21:00

Mathematics, 15.02.2021 21:00

Mathematics, 15.02.2021 21:00

Mathematics, 15.02.2021 21:00


Mathematics, 15.02.2021 21:00


Chemistry, 15.02.2021 21:00

Mathematics, 15.02.2021 21:00

Mathematics, 15.02.2021 21:00


Mathematics, 15.02.2021 21:00


Computers and Technology, 15.02.2021 21:00

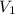 = 2 L,
= 2 L, 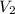 = 3 L,
= 3 L, 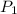 = X,
= X,  = 0.1 X,
= 0.1 X,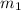 = 4.8 g,
= 4.8 g, 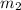 = 0.36 g,
= 0.36 g,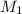 = ?,
= ?, 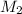 = ?
= ?