
Chemistry, 11.03.2020 02:25 unicornpoop54
A certain chemical reaction releases 480. kJ of heat energy per mole of reactant consumed. Suppose some moles of the reactant are put into a calorimeter (a device for measuring heat flow). It takes 3.85 J of heat energy to raise the temperature of this calorimeter by 1 °C. Now the reaction is run until all the reactant is gone, and the temperature of the calorimeter is found to rise by 14.6 °C. How would you calculate the number of moles of reactant that were consumed? Set the math up. But don't do any of it. Just leave your answer as a math expression. Also, be sure your answer includes all the correct unit symbols. moles consumed-

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1

Chemistry, 22.06.2019 09:30
The chart shows the bid provided by four contractors to complete a job. which contractor is the most cost-effective?
Answers: 3

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
A certain chemical reaction releases 480. kJ of heat energy per mole of reactant consumed. Suppose s...
Questions

Mathematics, 21.06.2019 19:00


Mathematics, 21.06.2019 19:00



English, 21.06.2019 19:00



Mathematics, 21.06.2019 19:00

History, 21.06.2019 19:00



Mathematics, 21.06.2019 19:00


Mathematics, 21.06.2019 19:00

Mathematics, 21.06.2019 19:00




History, 21.06.2019 19:00

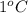 . So here, specific heat is given as 3.85 J. And, the change in temperature is
. So here, specific heat is given as 3.85 J. And, the change in temperature is 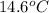 .
.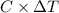
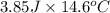
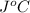
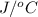 of heat is calculated as follows.
of heat is calculated as follows.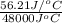
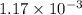 moles
moles

