
Chemistry, 11.03.2020 17:43 babbity2009
Hydroxylamine nitrate contains 29.17 mass % N, 4.20 mass % H, and 66.63 mass O. If its molar mass is between 94 and 98 g/mol, what is its molecular formula

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Five students had to answer the question how are elements arranged in a periodic tabledamon: i think the elements are arranged by increasing massflo: i think the elements are arranged according to their properties sienna: i think the elements are arranged by when their discovers kyle: i think the elements are arranged according to how common they areglenda: i don't agree with any of themwho is right
Answers: 1

Chemistry, 22.06.2019 03:00
Which best describes how johannes kepler developed his laws of planetary motion
Answers: 3

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 19:00
How many moles of cu are needed to react with 5.8 moles of agno3? cu + 2 agno3 → cu(no3)2 + 2 ag
Answers: 3
You know the right answer?
Hydroxylamine nitrate contains 29.17 mass % N, 4.20 mass % H, and 66.63 mass O. If its molar mass is...
Questions

Computers and Technology, 24.07.2020 19:01




English, 24.07.2020 19:01



Geography, 24.07.2020 19:01

World Languages, 24.07.2020 19:01


Mathematics, 24.07.2020 19:01


Mathematics, 24.07.2020 19:01


Mathematics, 24.07.2020 19:01





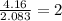 -----> H and O
-----> H and O
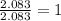 -----> N
-----> N 

