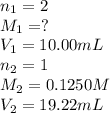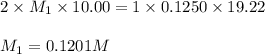Chemistry, 11.03.2020 18:35 genyjoannerubiera
You are using a standardized 0.1250 M solution of sodium hydroxide to determine the concentration of sulfuric acid. After pipetting 10.00 mL of the H2SO4 into your flask you find that you use 19.22 mL of your NaOH solution to reach the endpoint. No, wait... I mean What is the concentration of the sulfuric acid? H2SO4 + 2NaOH ---> Na2SO4 + 2HOH

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2

Chemistry, 22.06.2019 14:00
Which of the following is true about a carbonated soft drink? . the carbon dioxide is the solvent, and water is the solute.. the water is the solution, and carbon dioxide is the solvent.. the carbon dioxide is the solution, and the water is the solvent.. the water is the solvent, and the carbon dioxide is the solute.. .
Answers: 1

Chemistry, 22.06.2019 23:00
Which organism develops breathing organism develops breathing organs from pharyngeal arches? shark, spider, sea star, sea horse
Answers: 2
You know the right answer?
You are using a standardized 0.1250 M solution of sodium hydroxide to determine the concentration of...
Questions

Mathematics, 28.06.2019 03:00


Biology, 28.06.2019 03:00



Geography, 28.06.2019 03:00

Mathematics, 28.06.2019 03:00

Mathematics, 28.06.2019 03:00

Mathematics, 28.06.2019 03:00

English, 28.06.2019 03:00


History, 28.06.2019 03:00

Health, 28.06.2019 03:00


World Languages, 28.06.2019 03:00

Mathematics, 28.06.2019 03:00

Mathematics, 28.06.2019 03:00

History, 28.06.2019 03:00


Biology, 28.06.2019 03:00

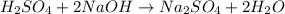
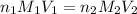
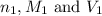 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 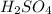
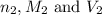 are the n-factor, molarity and volume of base which is NaOH.
are the n-factor, molarity and volume of base which is NaOH.