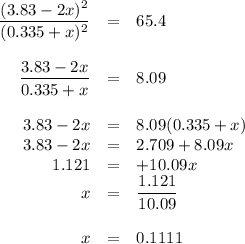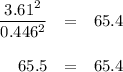Chemistry, 11.03.2020 22:26 gshreya2005
At 500 °C, hydrogen iodide decomposes according to 2 HI ( g ) − ⇀ ↽ − H 2 ( g ) + I 2 ( g ) For HI ( g ) heated to 500 °C in a 1.00 L reaction vessel, chemical analysis determined these concentrations at equilibrium: [ H 2 ] = 0.335 M , [ I 2 ] = 0.335 M , and [ HI ] = 2.83 M . If an additional 1.00 mol of HI ( g ) is introduced into the reaction vessel, what are the equilibrium concentrations after the new equilibrium has been reached?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 17:30
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 23.06.2019 03:50
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer
Answers: 1

Chemistry, 23.06.2019 15:40
Glucose (c6h12o6) is the simple sugar that plants make. what is the total number of atoms in glucose? 1 3 24 144
Answers: 1

Chemistry, 23.06.2019 17:00
Liquid nitrogen is kept at a temperature of -320 degrees. when liquid nitrogen is heated it quickly boils and turns into gas. which pair of pictures represent the change caused by adding heat to liquid nitrogen?
Answers: 3
You know the right answer?
At 500 °C, hydrogen iodide decomposes according to 2 HI ( g ) − ⇀ ↽ − H 2 ( g ) + I 2 ( g ) For HI (...
Questions


Mathematics, 02.12.2020 22:40

Mathematics, 02.12.2020 22:40


Mathematics, 02.12.2020 22:40





Health, 02.12.2020 22:40

Mathematics, 02.12.2020 22:40


History, 02.12.2020 22:40

Arts, 02.12.2020 22:40






![K_{\text{eq}} = \dfrac{\text{[HI]$^{2}$}}{\text{[H$_{2}]$[I$_{2}$]}} = \dfrac{2.83^{2}}{0.335 \times 0.335} = 65.4](/tpl/images/0543/5148/5b908.png)
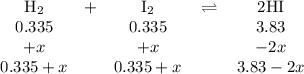
![K_{\text{c}} = \dfrac{\text{[HI]$^{2}$}}{\text{[H$_{2}$][I$_2$]}} = \dfrac{(3.83 - 2x)^{2}}{(0.335 + x)^{2}} = 65.4](/tpl/images/0543/5148/b1d26.png)