
Chemistry, 11.03.2020 22:56 amberpetty4288
The value of Ka for acetic acid , CH3COOH , is 1.80×10-5 . Write the equation for the reaction that goes with this equilibrium constant. (Use H3O+ instead of H+.)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 00:00
How many liters of water vapor can be produced if 108 grams of methane gas (ch4) are combusted at 312 k and 0.98 atm? show all work. pls ! will mark as brainliest
Answers: 1

Chemistry, 23.06.2019 00:30
Nuclear decay is the spontaneous decay of one element into a. an x-ray b. a ray of light c. another element
Answers: 1
You know the right answer?
The value of Ka for acetic acid , CH3COOH , is 1.80×10-5 . Write the equation for the reaction that...
Questions




Computers and Technology, 08.07.2019 18:20

Computers and Technology, 08.07.2019 18:20





Computers and Technology, 08.07.2019 18:20


Mathematics, 08.07.2019 18:20


Mathematics, 08.07.2019 18:20

English, 08.07.2019 18:20

Biology, 08.07.2019 18:20



Mathematics, 08.07.2019 18:20

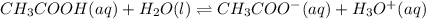
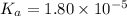
![K_c=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH][H_2O]}](/tpl/images/0543/5879/7c275.png)
![K_a=K_c\times [H_2O]=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH]}](/tpl/images/0543/5879/125a5.png)
![[H_2O]=1](/tpl/images/0543/5879/b8579.png)
![K_a=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH]}](/tpl/images/0543/5879/e4df0.png)


