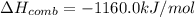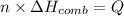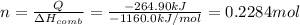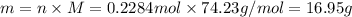Chemistry, 12.03.2020 01:59 cordovatierra16
If the heat of combustion for a specific compound is − 1160.0 kJ / mol and its molar mass is 74.23 g / mol, how many grams of this compound must you burn to release 264.90 kJ of heat?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 19:30
Awoman's basketball has a circumference between 28.5 and 29.0 inches and a maximum weight of 20 ounces (two significant figures). what are these specifications in units of centimeters and grams?
Answers: 2
You know the right answer?
If the heat of combustion for a specific compound is − 1160.0 kJ / mol and its molar mass is 74.23 g...
Questions


Chemistry, 20.09.2019 10:30

History, 20.09.2019 10:30

Biology, 20.09.2019 10:30


Arts, 20.09.2019 10:30

History, 20.09.2019 10:30




Mathematics, 20.09.2019 10:30

Mathematics, 20.09.2019 10:30

Mathematics, 20.09.2019 10:30



History, 20.09.2019 10:30

History, 20.09.2019 10:30

History, 20.09.2019 10:30