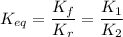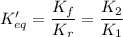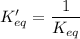Chemistry, 15.03.2020 23:43 babbybronx
The equilibrium-constant expression for a reaction written in one direction is the of the one for the reaction written for the reverse direction.

Answers: 3


Another question on Chemistry


Chemistry, 23.06.2019 07:30
Which of the following statements best explains why chemistry is testable a) it can measure data by experiments b) it cannot add new evidence c) it cannot be verified d) it is biased
Answers: 1

Chemistry, 23.06.2019 10:00
The image shows the process of which is used in nuclear power plants. photo attached
Answers: 1

Chemistry, 23.06.2019 12:30
What would happen to a weak base dissociation equilibrium if more products we added
Answers: 1
You know the right answer?
The equilibrium-constant expression for a reaction written in one direction is the of the one for t...
Questions



Biology, 08.12.2021 19:00


Mathematics, 08.12.2021 19:00





Mathematics, 08.12.2021 19:00


Mathematics, 08.12.2021 19:00

World Languages, 08.12.2021 19:00

Mathematics, 08.12.2021 19:00


History, 08.12.2021 19:00

Physics, 08.12.2021 19:00

Mathematics, 08.12.2021 19:00