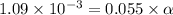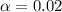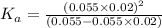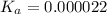Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 11:40
Consider this equilibrium: n29) + o2(g) + 2no(c).nitrogen gas and oxygen gas react when placed in a closed container. as the reaction proceeds towards equilibrium, what happens to the rate of thereverse reaction?
Answers: 1


Chemistry, 22.06.2019 21:50
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1
You know the right answer?
) PABA refers to para-aminobenzoic acid which is used in some sunscreen formulations. If a 0.055 M s...
Questions

Mathematics, 04.04.2021 20:10


Social Studies, 04.04.2021 20:10


Biology, 04.04.2021 20:10

Advanced Placement (AP), 04.04.2021 20:10

History, 04.04.2021 20:10

Mathematics, 04.04.2021 20:10

Mathematics, 04.04.2021 20:10


Mathematics, 04.04.2021 20:10


French, 04.04.2021 20:10

History, 04.04.2021 20:10


Social Studies, 04.04.2021 20:10




Chemistry, 04.04.2021 20:10

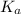 of PABA is 0.000022
of PABA is 0.000022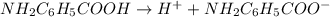
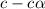
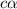
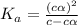
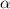 = ?
= ?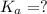
![pH=-log[H^+]](/tpl/images/0548/7597/15713.png)
![2.96=-log[H^+]](/tpl/images/0548/7597/5022f.png)
![[H^+]=1.09\times 10^{-3}](/tpl/images/0548/7597/3aec8.png)
![[H^+]=c\times \alpha](/tpl/images/0548/7597/4fc41.png)