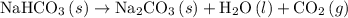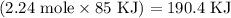Chemistry, 16.03.2020 21:15 msjsnell29
Given the following information: aHCO3(s)+85 kJ Na2CO3(s)+H2O(1) CO2(g) Calculate the amount of heat (ink) required to decompose 2.24 mol NaHCO3(s). You must show your work to receive credit .

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 22.06.2019 22:20
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2

Chemistry, 23.06.2019 00:00
What is the pressure of 0.500 moles of carbon dioxide gas in a 2.5 l tank and at a temperature of 301 k? (r=0.0821 l·atm/mol·k) 3.08 atm 1.2 atm 0.23 atm 4.01 atm 4.94 atm
Answers: 1

You know the right answer?
Given the following information: aHCO3(s)+85 kJ Na2CO3(s)+H2O(1) CO2(g) Calculate the amount of heat...
Questions

Biology, 24.11.2020 19:40






Mathematics, 24.11.2020 19:40

Mathematics, 24.11.2020 19:40


Health, 24.11.2020 19:40

Arts, 24.11.2020 19:40



Mathematics, 24.11.2020 19:40

Chemistry, 24.11.2020 19:40

Mathematics, 24.11.2020 19:40

Mathematics, 24.11.2020 19:40

Mathematics, 24.11.2020 19:40

Biology, 24.11.2020 19:40

Mathematics, 24.11.2020 19:40

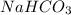 is shown below
is shown below