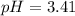Chemistry, 17.03.2020 00:57 claytonhopkins
A 1.50 L buffer solution is 0.250 M in HF and 0.250 M in NaF. Calculate the pH of the solution after the addition of 0.100 moles of solid NaOH. Assume no volume change upon the addition of base. The Ka for HF is 6.8 × 10-4.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:40
Achange in the number of neutrons in an atom will change an blank . when the number of protons changes in an atom, a new element will form.
Answers: 2


Chemistry, 22.06.2019 19:30
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2

Chemistry, 22.06.2019 20:00
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2
You know the right answer?
A 1.50 L buffer solution is 0.250 M in HF and 0.250 M in NaF. Calculate the pH of the solution after...
Questions


Chemistry, 31.03.2020 03:41

Mathematics, 31.03.2020 03:41





Mathematics, 31.03.2020 03:41




Mathematics, 31.03.2020 03:41

Computers and Technology, 31.03.2020 03:41







Mathematics, 31.03.2020 03:41

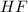 .
.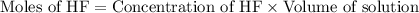
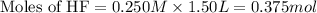
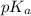 .
.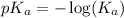
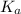 in this expression, we get:
in this expression, we get: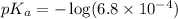
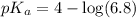
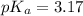
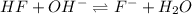
![pH=pK_a+\log \frac{[Salt]}{[Acid]}](/tpl/images/0549/7799/e961a.png)
![pH=pK_a+\log \frac{[F^-]}{[HF]}](/tpl/images/0549/7799/bef2f.png)
![pH=3.17+\log [\frac{(\frac{0.475}{1.50})}{(\frac{0.275}{1.50})}]](/tpl/images/0549/7799/a4ae7.png)