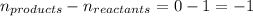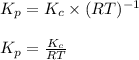Chemistry, 17.03.2020 02:00 meowmeowcow
Consider the following chemical equilibrium: 2PbO+O2=2PbO2Now write an equation below that shows how to calculate from for this reaction at an absolute temperature . You can assume is comfortably above room temperature. If you include any common physical constants in your equation be sure you use their standard symbols, found in the ALEKS Calculator.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 05:30
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1

Chemistry, 22.06.2019 23:00
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2
You know the right answer?
Consider the following chemical equilibrium: 2PbO+O2=2PbO2Now write an equation below that shows how...
Questions


Computers and Technology, 20.07.2019 02:20

Medicine, 20.07.2019 02:20



Medicine, 20.07.2019 02:20

Computers and Technology, 20.07.2019 02:20

Medicine, 20.07.2019 02:20

Medicine, 20.07.2019 02:20

Medicine, 20.07.2019 02:20

Engineering, 20.07.2019 02:20

Medicine, 20.07.2019 02:30


Medicine, 20.07.2019 02:30





Engineering, 20.07.2019 02:30

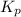 from
from 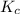 for this reaction at an absolute temperature T.
for this reaction at an absolute temperature T.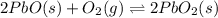
![K_c=\frac{1}{[O_2]}](/tpl/images/0549/9223/ce11c.png)
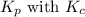 is given by the formula:
is given by the formula: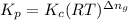
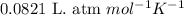
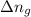 = change in number of moles of gas particles =
= change in number of moles of gas particles =