
Chemistry, 17.03.2020 03:18 violetagamez2
Sodium sulfate is slowly added to a solution containing 0.0500 M Ca 2 + ( aq ) and 0.0300 M Ag + ( aq ) . What will be the concentration of Ca 2 + ( aq ) when Ag 2 SO 4 ( s ) begins to precipitate? Solubility-product constants, K sp , can be found in the chempendix.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Achef makes salad dressing by mixing oil, vinegar, and spices, as shown. which type of matter is the salad dressing?
Answers: 1

Chemistry, 22.06.2019 02:30
24 points and brainliest to anyone who can answer under 10 minutes with best ! the table below shows the role of different substances during photosynthesis. substance role during photosynthesis glucose stores chemical energy water combines with glucose to form carbon dioxide chlorophyll traps sunlight which of the following statements would correct one of the roles listed in the table? glucose combines with carbon to form water. chlorophyll reacts with light to produce carbon dioxide. water combines with carbon dioxide during photosynthesis. chlorophyll stores chemical energy needed for photosynthesis.
Answers: 1


You know the right answer?
Sodium sulfate is slowly added to a solution containing 0.0500 M Ca 2 + ( aq ) and 0.0300 M Ag + ( a...
Questions


Mathematics, 23.03.2021 21:30



English, 23.03.2021 21:30

Computers and Technology, 23.03.2021 21:30


Mathematics, 23.03.2021 21:30







English, 23.03.2021 21:30




English, 23.03.2021 21:30

Mathematics, 23.03.2021 21:30

![[Ca^{2+}]](/tpl/images/0550/0535/17576.png) ion is, 0.00371 M
ion is, 0.00371 M![[SO_4^{2-}]](/tpl/images/0550/0535/43b69.png) ion.
ion.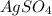 will be:
will be: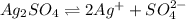
![K_{sp}=[Ag^{+}]^2[SO_4^{2-}]](/tpl/images/0550/0535/80963.png)

![1.20\times 10^{-5}=(0.0300)^2\times [SO_4^{2-}]](/tpl/images/0550/0535/d8931.png)
![[SO_4^{2-}]=0.0133M](/tpl/images/0550/0535/cd404.png)
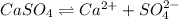
![K_{sp}=[Ca^{2+}][SO_4^{2-}]](/tpl/images/0550/0535/958f2.png)
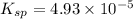
![4.93\times 10^{-5}=[Ca^{2+}]\times (0.0133)](/tpl/images/0550/0535/7fa1a.png)
![[Ca^{2+}]=0.00371M](/tpl/images/0550/0535/7d329.png)



