
Chemistry, 17.03.2020 04:05 lilrel8602
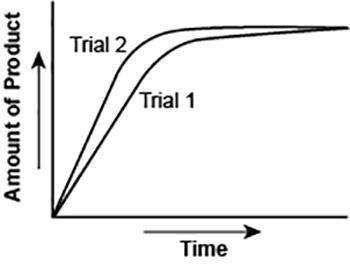
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different concentration of reactant was used during each trial, whereas the other factors were kept constant.
Which of the following statements explains which trial has a lower concentration of the reactant?
A. Trial 1, because the average rate of the reaction is lower.
B. Trial 1, because this reaction lasted for a longer duration than Trial 2.
C. Trial 2, because this reaction was initially fast and later slowed down.
D. Trial 2, because the volume of product formed per unit time was higher.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 10:30
Skills of homo sapiens were found an excavation. the skulls were preserved because the bodies were frozen. so, these fossils are (blank) fossils.the image shows the evolution of skulls beginning 2 to 3 million years ago. based on the image, modern human skulls(blank) ape skulls.
Answers: 1

Chemistry, 23.06.2019 04:20
The graph shows one consequence of urban sprawl. how did urban sprawl contribute to the change in biodiversity
Answers: 2

Chemistry, 23.06.2019 06:00
Each step in the following process has a yield of 70% ch4 + 4cl2 yield ccl4 +4hcl ccl4 + 2hf yield ccl2f2 + 2hcl of 4.50 mole ch4 reacts what is the total amount of hcl produced
Answers: 3
You know the right answer?
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different...
Questions


Mathematics, 26.01.2021 03:20

Mathematics, 26.01.2021 03:20


Mathematics, 26.01.2021 03:20


Chemistry, 26.01.2021 03:20




Mathematics, 26.01.2021 03:20


Geography, 26.01.2021 03:20

Mathematics, 26.01.2021 03:20

English, 26.01.2021 03:20



Mathematics, 26.01.2021 03:20





