
Chemistry, 17.03.2020 04:31 lobatospitones
Determine the rate constant for each of the following fi rst-order reactions, in each case expressed for the rate of loss of A: (a) A S B, given that the concentration of A decreases to one-half its initial value in 1000. s; (b) A S B, given that the concentration of A decreases from 0.67 molL1 to 0.53 molL1 in 25 s; (c) 2 A S B C, given that [A]0 0.153 molL1 and that after 115 s the concentration of B rises to 0.034 molL1

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1

Chemistry, 22.06.2019 03:20
What is the ima of the 1 st class lever in the graphic given? 2 3 0.5
Answers: 1

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

You know the right answer?
Determine the rate constant for each of the following fi rst-order reactions, in each case expressed...
Questions


History, 02.07.2021 01:00


Mathematics, 02.07.2021 01:00




History, 02.07.2021 01:00


Mathematics, 02.07.2021 01:00










![[A]=[A]_o\times e^{-k\times t}](/tpl/images/0550/2081/d7033.png)
![[A]_o](/tpl/images/0550/2081/9caf5.png) = initial concentration of reactant
= initial concentration of reactant ![[A_o]=x](/tpl/images/0550/2081/aecde.png)
![[A]=\frac{x}{2}](/tpl/images/0550/2081/f7c20.png)
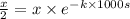
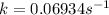
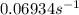 .
.![[A_o]=0.67 mol/L](/tpl/images/0550/2081/74ad9.png)
![[A]=0.53 mol/L](/tpl/images/0550/2081/d3d0a.png)
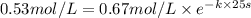
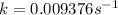
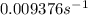 .
.![[A_o]=0.153 mol/L](/tpl/images/0550/2081/3406c.png)
![[A]=?](/tpl/images/0550/2081/8be21.png)
![[B]=0.034 mol/L](/tpl/images/0550/2081/2fbaf.png)
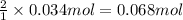 of A
of A![[A]=0.153 mol/L-0.068 mol/L=0.085 mol/L](/tpl/images/0550/2081/97bbf.png)
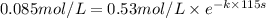
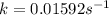
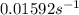 .
.

