
Chemistry, 17.03.2020 05:59 milkshakegrande101
Balance the chemical equation given below, and calculate the volume of nitrogen monoxide gas produced when 8.00 g of ammonia is reacted with 12.0 g of oxygen at 25°C? The density of nitrogen monoxide at 25°C is 1.23 g/L. ___ NH3(g) + ___ O2(g) → ___ NO(g) + ___ H2O(l)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:30
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1

Chemistry, 22.06.2019 23:00
What does a numerical subscript following an element in a chemical formula mean?
Answers: 1

Chemistry, 23.06.2019 00:00
How many peaks will be present in a mass spectrum for brcl?
Answers: 1
You know the right answer?
Balance the chemical equation given below, and calculate the volume of nitrogen monoxide gas produce...
Questions




Mathematics, 28.01.2020 09:31


Health, 28.01.2020 09:31



Mathematics, 28.01.2020 09:31

English, 28.01.2020 09:31



Mathematics, 28.01.2020 09:31

Mathematics, 28.01.2020 09:31




History, 28.01.2020 09:31


Biology, 28.01.2020 09:31

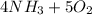 →
→ 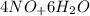
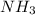 here acts as an limiting reagent.
here acts as an limiting reagent. 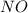
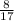 = 0.470 mol
= 0.470 mol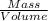
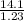 = 11.46 lit
= 11.46 lit 

