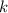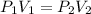A certain amount of chlorine gas was placed inside a cylinder with a movable piston at one end. The initial volume was 3.00 L and the initial pressure of chlorine was 1.85 atm . The piston was pushed down to change the volume to 1.00 L. Calculate the final pressure of the gas if the temperature and number of moles of chlorine remain constant

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:30
What metric units would you use to measure the thickness of a key
Answers: 3

Chemistry, 23.06.2019 01:30
What is produced from neutralization of an acid and a base? a. hydronium ions b. citric acid c. salt and water
Answers: 1

Chemistry, 23.06.2019 06:30
What is the chemical formula for a compound between li and br? libr li2br libr2 libr3
Answers: 1

Chemistry, 23.06.2019 07:00
Achemist who studies water samples did a demonstration of how to test for lead in water. she added a clear solution of potassium iodide to a clear solution of lead nitrate. then a yellow swirling solid formed in the liquid. what is most likely true about the yellow solid?
Answers: 3
You know the right answer?
A certain amount of chlorine gas was placed inside a cylinder with a movable piston at one end. The...
Questions


English, 20.09.2020 08:01


English, 20.09.2020 08:01



Computers and Technology, 20.09.2020 08:01



History, 20.09.2020 08:01

Mathematics, 20.09.2020 08:01

Engineering, 20.09.2020 08:01


Biology, 20.09.2020 08:01

Mathematics, 20.09.2020 08:01





Mathematics, 20.09.2020 08:01

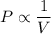
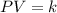 Pressure multiplied by volume equals some constant
Pressure multiplied by volume equals some constant