Chemistry, 18.03.2020 18:57 dyllanmasters99
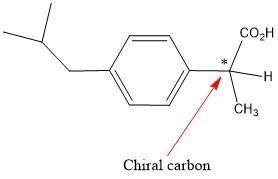
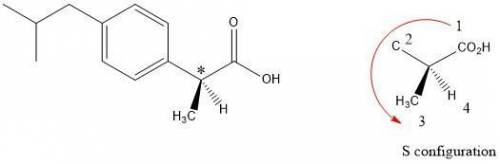
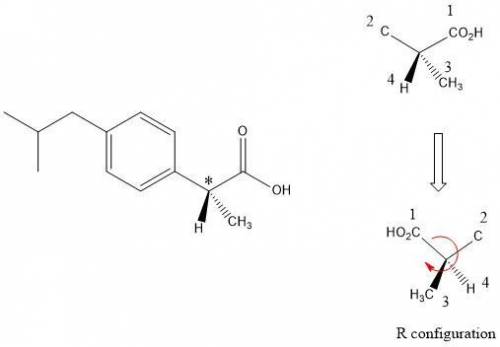
Buprofen, a well-known non-steroidal anti-inflammatory drug, has chirality. Only the S enantiomer has anti-inflammatory activity (although the R enantiomer is converted slowly by the body into the S enantiomer). Add wedge-and-dash bonds to complete the perspective structures of the two stereoisomers of ibuprofen.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:50
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1

Chemistry, 22.06.2019 17:00
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 22.06.2019 23:00
What is the oxidation state of each individual carbon atom in c2o42−?
Answers: 1
You know the right answer?
Buprofen, a well-known non-steroidal anti-inflammatory drug, has chirality. Only the S enantiomer ha...
Questions



Mathematics, 09.11.2020 20:20


Business, 09.11.2020 20:20




Health, 09.11.2020 20:20


Mathematics, 09.11.2020 20:20


English, 09.11.2020 20:20

Health, 09.11.2020 20:20



Social Studies, 09.11.2020 20:20

Social Studies, 09.11.2020 20:20

English, 09.11.2020 20:20

Mathematics, 09.11.2020 20:20