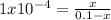Chemistry, 18.03.2020 19:39 JellalFernandes
A reaction A(g)⇌B(g)has an equilibrium constant of 1.0×10−4 For which of the initial reaction mixtures is the small approximation most likely to apply? Explain why.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:00
If 1.63 times 10 negative 4 of helium dissolves in 100.0g of water, what is the concentration in parts per million
Answers: 3

Chemistry, 23.06.2019 03:30
Which of the following describes the entropy change as a solution is made from a liquid and solid
Answers: 1

Chemistry, 23.06.2019 04:00
Calculate the mass of 0.750 mol of the following substance. na3po4. , i'm not quite sure on how to set up the problem to solve! : (
Answers: 1

Chemistry, 23.06.2019 12:30
How does a nuclear reactor produce electricity? a. high-energy gamma rays are converted by a generator into electricity. b. the heat from the reaction turns water to steam, which runs a generator. c. the reaction produces a stream of electrons that flow through wires and into batteries. d. the heat released from the reaction is used to burn coal or gas to produce electricity. e. control rods absorb the neutrons emitted and release a stream of electrons as electricity.
Answers: 1
You know the right answer?
A reaction A(g)⇌B(g)has an equilibrium constant of 1.0×10−4 For which of the initial reaction mixtur...
Questions



English, 01.03.2021 04:40

English, 01.03.2021 04:40

History, 01.03.2021 04:40

Computers and Technology, 01.03.2021 04:40



Mathematics, 01.03.2021 04:40


Mathematics, 01.03.2021 04:40

Mathematics, 01.03.2021 04:40

Mathematics, 01.03.2021 04:40



Mathematics, 01.03.2021 04:40



Mathematics, 01.03.2021 04:40

 in this case would be 0.00001M which is the smallest one from the given options, considering:
in this case would be 0.00001M which is the smallest one from the given options, considering: