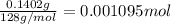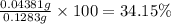Chemistry, 18.03.2020 21:26 Carlosanddana123
You are given a sample of limestone, which is mostly CaCO3, to determine the mass percentage of Ca in the rock. You dissolve the limestone in hydrochloric acid, which gives a solution of calcium chloride. Then you precipitate the calcium ion in solution by adding sodium oxalate, Na2C2O4. The precipitate is calcium oxalate, CaC2O4. You find that a sample of limestone weighing 128.3 mg gives 140.2 mg of CaC2O4. What is the mass percentage of calcium in the limestone

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

You know the right answer?
You are given a sample of limestone, which is mostly CaCO3, to determine the mass percentage of Ca i...
Questions



English, 09.12.2020 23:10


Chemistry, 09.12.2020 23:10



History, 09.12.2020 23:10

History, 09.12.2020 23:10


Mathematics, 09.12.2020 23:10


Spanish, 09.12.2020 23:10

Chemistry, 09.12.2020 23:10

Chemistry, 09.12.2020 23:10

Mathematics, 09.12.2020 23:10


Mathematics, 09.12.2020 23:10


Mathematics, 09.12.2020 23:10