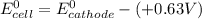A certain half-reaction has a standard reduction potential E⁰red = +0.63V. An engineer proposes using this half-reaction at the cathode of a galvanic cell that must provide at least 1.30V of electrical power. The cell will operate under standard conditions.
(a) Is there a minimum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, write "yes" and calculate the minimum. Round your answer to 2 decimal places. If there is no lower limit, write "no".
(b) Is there a maximum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, write "yes" and calculate the minimum. Round your answer to 2 decimal places. If there is no upper limit, write "no".

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
How to solve 4 nh3(g) + 5 o2(g) > 4 no(g) + 6 h2o(g) in chemistry
Answers: 1


Chemistry, 22.06.2019 23:00
What is the average rate of the reaction between 10 and 20 s?
Answers: 1

Chemistry, 23.06.2019 03:50
How many liters of oxygen gas, at standardtemperature and pressure, will react with 35.8 grams ofiron metal? 4 fe (s) + 3 o2 (g) → 2 fe2o3 (s)
Answers: 3
You know the right answer?
A certain half-reaction has a standard reduction potential E⁰red = +0.63V. An engineer proposes usin...
Questions


Mathematics, 16.01.2020 07:31



World Languages, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31

History, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31




Health, 16.01.2020 07:31

Chemistry, 16.01.2020 07:31


Computers and Technology, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31


English, 16.01.2020 07:31


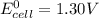
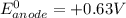
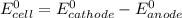
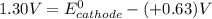
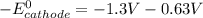
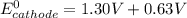
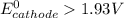 ; then
; then 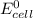 > 1.30 V
> 1.30 V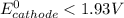 ; then
; then 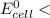 1.30 V
1.30 V