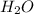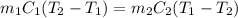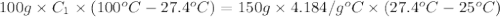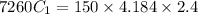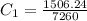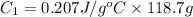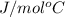A 100.0g sample of tin is heated to 100.0 oC (Celsius) and is placed in a coffee cup calorimeter containing 150. g of water at 25.0 oC. After the metal cools, the final temperature of the metal and the water is 27.4 oC. Calculate the specific heat capacity of tin from these experimental data, assuming that no heat escapes to the surroundings or is transferred to the calorimeter. Specific heat of water

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3



Chemistry, 22.06.2019 19:40
What is the wavelength of a 3*10^12 hz infrared wave a 3*10^20m b 1* 10^4m c 3*10^-3m d 1*10^-4 m
Answers: 1
You know the right answer?
A 100.0g sample of tin is heated to 100.0 oC (Celsius) and is placed in a coffee cup calorimeter con...
Questions

Mathematics, 07.10.2020 14:01

Biology, 07.10.2020 14:01



Mathematics, 07.10.2020 14:01

Mathematics, 07.10.2020 14:01

Business, 07.10.2020 14:01

Mathematics, 07.10.2020 14:01

Arts, 07.10.2020 14:01

Mathematics, 07.10.2020 14:01

Computers and Technology, 07.10.2020 14:01


Mathematics, 07.10.2020 14:01

History, 07.10.2020 14:01

English, 07.10.2020 14:01




Mathematics, 07.10.2020 14:01

English, 07.10.2020 14:01

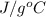 and atomic mass of tin is 118.7 g/mol. For the given situation,
and atomic mass of tin is 118.7 g/mol. For the given situation, 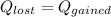
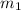 = mass of Sn
= mass of Sn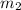 = mass of
= mass of