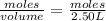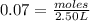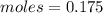Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 22.06.2019 18:00
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1

Chemistry, 23.06.2019 08:00
If the solubility of a gas in water is 1.22 g/l at 2.75 atm, what is its solubility (in g/l) at 1.0 atm?
Answers: 1

Chemistry, 23.06.2019 10:10
Which orbitals form a pi bond? a. the s orbital and three p orbitals b. the s orbital and two p orbitals c. overlapping p orbitals d. overlapping hybrid orbitals
Answers: 2
You know the right answer?
10. At a certain temperature, Kc equals 1.4 × 102 for the reaction:2 CO(g) + O2(g) ⇌ 2 CO2(g). If a...
Questions




English, 24.04.2020 22:37



Computers and Technology, 24.04.2020 22:37


English, 24.04.2020 22:37



Mathematics, 24.04.2020 22:37


English, 24.04.2020 22:37

English, 24.04.2020 22:37

Spanish, 24.04.2020 22:37




Mathematics, 24.04.2020 22:37

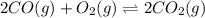
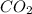 =
= 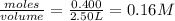
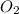 =
= 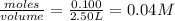
![K_c=\frac{[CO_2]^2}{[CO]^2\times [O_2]}](/tpl/images/0553/0997/662e3.png)
![1.4\times 10^2=\frac{(0.16)^2}{[CO]^2\times 0.04}](/tpl/images/0553/0997/5df84.png)
![[CO]=0.07M](/tpl/images/0553/0997/06724.png)
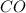 =
=