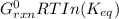Chemistry, 19.03.2020 03:02 nadinealonzo6121
A critical reaction in the production of energy to do work or drive chemical reactions in biological systems is the hydrolysis of adenosine triphosphate, ATP, to adenosine diphosphate, ADP, as described by the reaction ATP ( aq ) + H 2 O ( l ) ⟶ ADP ( aq ) + HPO 2 − 4 ( aq ) for which Δ G ∘ rxn = − 30.5 kJ/mol at 37.0 °C and pH 7.0. Calculate the value of Δ G rxn in a biological cell in which [ ATP ] = 5.0 mM, [ ADP ] = 0.20 mM, and [ HPO 2 − 4 ] = 5.0 mM.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 21.06.2019 19:00
Which term best describes the form sound takes as it travels away from a drum (a- gas)(b-music) ( c-waves) (d-particles
Answers: 3

Chemistry, 22.06.2019 00:30
Used the balanced equation 2h2+ o2 - -> 2h2o. if you have 7.2 grams of o2 , how many grams of h2o can you produce ?
Answers: 2

Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1
You know the right answer?
A critical reaction in the production of energy to do work or drive chemical reactions in biological...
Questions




Mathematics, 30.10.2020 16:20


Mathematics, 30.10.2020 16:20

English, 30.10.2020 16:20






Physics, 30.10.2020 16:20






Mathematics, 30.10.2020 16:20

Mathematics, 30.10.2020 16:20


![K_{eq}= \frac{[ADP][HPO_4^{2-}]}{[ATP]}](/tpl/images/0553/4313/cfc41.png)
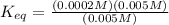
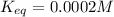
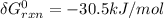 to J/mol; we have:
to J/mol; we have: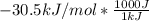
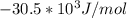
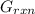 can be calculated as:
can be calculated as: