
A piece of iron (mass = 25.0 g) at 398 K is placed in a styrofoam coffee cup containing 25.0 mL of water at 298 K. Assuming that no heat is lost to the cup or the surroundings, what will the final temperature of the water be? The specific heat capacity of iron = 0.449 J/g°C and water = 4.18 J/g°C.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:00
In a closed or isolated system , matter can not be created or destroyed. it can change forms but is conserved true or false
Answers: 1

Chemistry, 21.06.2019 22:00
Bohr's model could only explain the spectra of which type of atoms? single atoms with one electron single atoms with more than one electron bonded atoms with one electron bonded atoms with more than one electron
Answers: 2

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3
You know the right answer?
A piece of iron (mass = 25.0 g) at 398 K is placed in a styrofoam coffee cup containing 25.0 mL of w...
Questions


History, 16.10.2020 08:01

Biology, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01


Biology, 16.10.2020 08:01




English, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01

History, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01

World Languages, 16.10.2020 08:01

History, 16.10.2020 08:01

Social Studies, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01

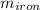 = 25 gm
= 25 gm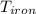 = 398 K
= 398 K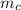 = 25 ml = 25 gm
= 25 ml = 25 gm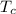 = 298 K
= 298 K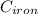 = 0.449
= 0.449 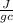
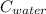 = 4.18
= 4.18 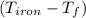 =
= 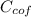 ×
× 
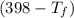 = 25 × 4.18 ×
= 25 × 4.18 × 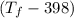
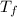 = 9.3 (
= 9.3 ( 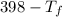 = 9.3
= 9.3 

