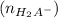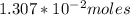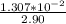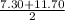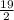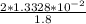Chemistry, 19.03.2020 07:14 yesseniaroman21
In the titration of 238.0 mL of a 5.60×10-2 M solution of acid H3A (Ka1 = 1.0×10-3, Ka2 = 5.0×10-8, Ka3 = 2.0×10-12), calculate the volume of 2.90 M NaOH required to reach the following pH values. pH = 9.50 Volume required = mL pH = 4.70 Volume required = mL

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1

Chemistry, 23.06.2019 01:30
How does the attraction between particles affect the ability of a solvent to dissolve in a substance
Answers: 1

Chemistry, 23.06.2019 02:30
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1

Chemistry, 23.06.2019 03:00
A0.100-kilogram apple hangs in a tree 1.50 meter above the ground. ignore frictional effects, the total mechanical energy of the apples is
Answers: 1
You know the right answer?
In the titration of 238.0 mL of a 5.60×10-2 M solution of acid H3A (Ka1 = 1.0×10-3, Ka2 = 5.0×10-8,...
Questions

Mathematics, 30.09.2019 12:50


English, 30.09.2019 12:50




Health, 30.09.2019 13:00

Geography, 30.09.2019 13:00




Mathematics, 30.09.2019 13:00

Mathematics, 30.09.2019 13:00

English, 30.09.2019 13:00



History, 30.09.2019 13:00

Chemistry, 30.09.2019 13:00


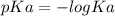
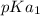 ; we have
; we have 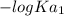
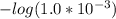
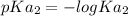
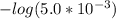
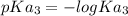
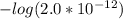
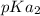 and
and 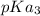 .
.![[H_3O^+]](/tpl/images/0553/6901/6cfd2.png) = pH = 4.70
= pH = 4.70![[H_3O^+] = 10^{-4.70}](/tpl/images/0553/6901/8c265.png)
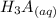

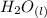 ⇄
⇄ 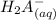
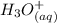
![Ka_1= \frac{[H_2A^-][H_3O^+]}{[H_3A]}](/tpl/images/0553/6901/4cf6a.png)
![1.0*10^{-3}= \frac{[H_2A^-][H_3O^+]}{[H_3A]}](/tpl/images/0553/6901/72bac.png)
![\frac{1.0*10^{-3}}{[H_3O^+]}= \frac{[H_2A^-]}{[H_3A]}](/tpl/images/0553/6901/eb284.png)
![\frac{1.0*10^{-3}}{[10^{-4.70}]}= \frac{[H_2A^-]}{[H_3A]}](/tpl/images/0553/6901/c148a.png)
![\frac{[H_2A^-]}{[H_3A]}= 10^{1.7}](/tpl/images/0553/6901/e4f78.png)
![\frac{[H_2A^-]}{[H_3A]}= 50.12](/tpl/images/0553/6901/480fb.png)
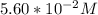 solution is given:
solution is given: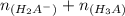 =
= 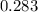
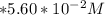
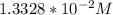
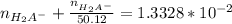
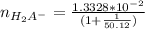
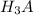 to
to 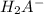 i.e
i.e