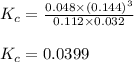Chemistry, 19.03.2020 07:08 xMABRYx1991
Steam reforming of methane ( ) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. An industrial chemist studying this reaction fills a tank with of methane gas and of water vapor at . He then raises the temperature, and when the mixture has come to equilibrium measures the amount of hydrogen gas to be . Calculate the concentration equilibrium constant for the steam reforming of methane at the final temperature of the mixture. Round your answer to significant digits.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1


Chemistry, 23.06.2019 03:30
The molar mass of nickel(ni) is 58.7 g/mol. how many moles are in an 88 gram sample of nickel?
Answers: 1
You know the right answer?
Steam reforming of methane ( ) produces "synthesis gas," a mixture of carbon monoxide gas and hydrog...
Questions




Mathematics, 21.10.2020 16:01


Computers and Technology, 21.10.2020 16:01


Health, 21.10.2020 16:01


Computers and Technology, 21.10.2020 16:01





Chemistry, 21.10.2020 16:01


Mathematics, 21.10.2020 16:01

Mathematics, 21.10.2020 16:01


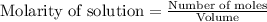
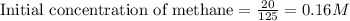
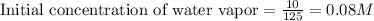
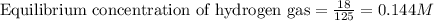
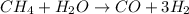
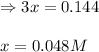
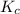 for above equation follows:
for above equation follows:![K_c=\frac{[CO][H_2]^3}{[CH_4][H_2O]}](/tpl/images/0553/6848/498b4.png)