
Chemistry, 19.03.2020 08:58 jetblackcap
Phosphorus pentachloride decomposes according to the chemical equation PCl 5 ( g ) − ⇀ ↽ − PCl 3 ( g ) + Cl 2 ( g ) K c = 1.80 at 250 ∘ C A 0.1846 mol sample of PCl 5 ( g ) is injected into an empty 2.55 L reaction vessel held at 250 ∘ C. Calculate the concentrations of PCl 5 ( g ) and PCl 3 ( g ) at equilibrium.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 13:00
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2

Chemistry, 22.06.2019 13:30
Apush or pull that moves or changes and object when to objects touch
Answers: 2

Chemistry, 22.06.2019 23:00
What is the oxidation state of an individual bromine atom in nabro3?
Answers: 2
You know the right answer?
Phosphorus pentachloride decomposes according to the chemical equation PCl 5 ( g ) − ⇀ ↽ − PCl 3 ( g...
Questions



English, 08.12.2021 08:20



Mathematics, 08.12.2021 08:20


Mathematics, 08.12.2021 08:20


English, 08.12.2021 08:20


Mathematics, 08.12.2021 08:20

Mathematics, 08.12.2021 08:20



Chemistry, 08.12.2021 08:20




English, 08.12.2021 08:20

![[PCl_3]_{eq}=0.0697M\\](/tpl/images/0553/8714/e4a7f.png)
![[PCl_5]_{eq}=0.00269M](/tpl/images/0553/8714/6813e.png)
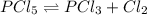
![[PCl_5]_0=\frac{0.1846mol}{2.55L}=0.0724M](/tpl/images/0553/8714/c9006.png)
![Kc=\frac{[Cl_2]_{eq}[PCl_3]_{eq}}{[PCl_5]_{eq}}](/tpl/images/0553/8714/38534.png)
 due to the reaction extent, it becomes:
due to the reaction extent, it becomes: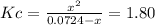

![[PCl_3]_{eq}=x=0.0697M\\](/tpl/images/0553/8714/b7f0d.png)
![[PCl_5]_{eq}=0.0724M-x=0.0724M-0.0697M=0.00269M](/tpl/images/0553/8714/fc102.png)


