
The decomposition of ozone in the upper atmosphere to dioxygen occurs by a two-step mechanism. The first step is a fast reversible step and the second is a slow reaction between an oxygen atom and an ozone molecule:Step 1: O3(g) O2(g) + O(g) Fast, reversible, reactionStep 2: O3(g) + O(g) → 2O2(g) SlowWhich is the rate determining step?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1

Chemistry, 23.06.2019 02:40
How can a mixture of salt water be separated into salt and water
Answers: 1
You know the right answer?
The decomposition of ozone in the upper atmosphere to dioxygen occurs by a two-step mechanism. The f...
Questions

History, 19.08.2019 12:00

Social Studies, 19.08.2019 12:00

Physics, 19.08.2019 12:00

Mathematics, 19.08.2019 12:00


Chemistry, 19.08.2019 12:00

Mathematics, 19.08.2019 12:00

English, 19.08.2019 12:00

Mathematics, 19.08.2019 12:00




Mathematics, 19.08.2019 12:00


Geography, 19.08.2019 12:00




Physics, 19.08.2019 12:00

History, 19.08.2019 12:00

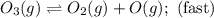
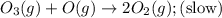
![\text{Rate}=k[O_3][O]](/tpl/images/0554/3771/ba34f.png)


